 
The Power of Atomic Numberįor more than half a century, chemists lived in an awkward space concerning the periodic table. Shortly after the publication of Mendeleev’s first table, it became clear that the table needed rearrangement. Tellurium has a heavier atomic weight than iodine, which forced Mendeleev to confusingly place it one space before iodine to maintain the chemical groupings. However, he knew that tellurium had similar chemical behavior to the oxygen group of elements. Mendeleev had arranged the table so that these similar chemical groups shared the same row, including a row for these halogens. It was the arrangement of this first group, termed “ halogens,” that raised the eyebrows of chemists. Chemists also grouped carbon, silicon, and selenium, as well as nitrogen, phosphorus, and arsenic for their similar behavior. Sodium, potassium, rubidium, and cesium had incredibly unstable neutral states and only seemed to form +1 charged ions. Fluorine, chlorine, bromine, and iodine each had diatomic elemental forms, as well as a strong propensity to exclusively ionize to a -1 charge. Chemists understood at the time that certain elements with very distant atomic masses had similar chemical characteristics. Second, and more troubling, the chemical behavior of the elements undermined mass-based ordering. Non-unique atomic masses suggested that it was impossible to meaningfully order elements in this way. At the time of the table’s formulation, chemists estimated the atomic masses of nickel and cobalt to roughly equal each other. First, some elements don’t have unique atomic masses. However, some quick observations of the table revealed that ordering the elements by atomic mass proved unhelpful and misleading. Mendeleev’s 1869 periodic table with elements arranged by atomic mass. Indeed, Mendeleev’s first periodic table arranges elements in a similar order to the modern table. Because atomic mass essentially equals the sum of protons and neutrons, it correlates strongly with atomic number. Dmitri Mendeleev, the architect of the modern periodic table, arranged his first table in 1869 according to atomic mass instead. In truth, chemists didn’t always use atomic numbers to categorize elements. The answer lies in the chemistry of different elements. 
Since we know atoms can vary not just in proton number, but also in neutron and electron number, why do we care so much about protons? After all, chemists organize elements by atomic number in the periodic table, which suggests some inherent importance tied to an atom’s proton number. Variants of an element with different electron numbers, and thus different electric charges, are called “ions” of an element. Also, oxygen has 8 electrons in its elemental form, but can also have 10 electrons given certain conditions. Variants of an element with different neutron numbers, and thus different atomic weights, are called “ isotopes” of the element. With carbon specifically in mind, its atomic structure most often has 6 neutrons, though chemists know about other forms of carbon with 7, 8, or more neutrons. An atom can have any number of neutrons and electrons, but as long as it has 6 protons, chemists will always consider it a carbon atom. Importantly, chemists use atomic number as the defining characteristic of an element. Thus, hydrogen atoms have 1 proton, carbon atoms have 6 protons, and so on. With this in mind, an element’s atomic number represents the number of protons found in one atom of the element. Nuclear atomic model with protons, neutrons, and electrons. Different elements involve atoms with different numbers of these subatomic particles. Atoms, themselves, are small round structures composed of what chemists call subatomic particles, namely protons, electrons, and neutrons. In essence, an element is a type of atom. However, atomic number isn’t arbitrary it says something fundamental about the subatomic structure of each element. You might even think of some data organization advantages that such numerical identifications would offer. Since there exist no gaps in atomic numbers from 1 to 118, it’s easy to presume atomic number only serves as some convenient numerical identification. On first blush, you might assume that an element’s atomic number is arbitrary. Modern periodic table with atomic numbers listed above atom symbols. 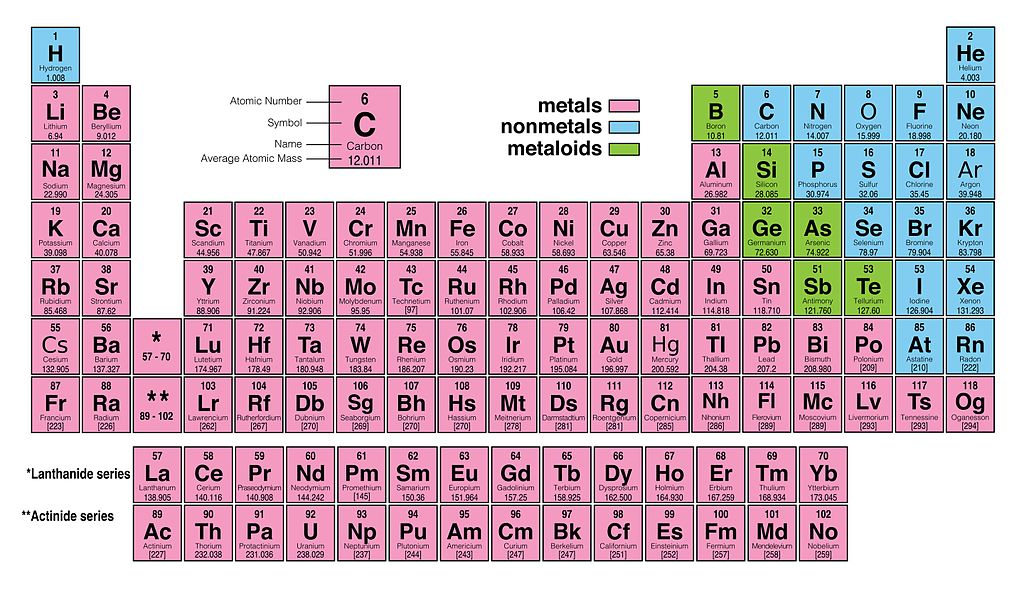
If you look at a periodic table, you will notice that each element has a unique value between 1 and 118 which chemists call “atomic number.” Hydrogen has an atomic number of 1. What exactly makes one element different from another? Why are carbon and hydrogen and oxygen considered to be different substances? What can we specifically point to that explains the difference between these elements on its most basic level? In this article, you will learn about atomic number, its definition, its usefulness in categorizing elements, and its history as a theory in chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
